1. Describe the three phases of matter in terms of shape and volume.
Solid matter has molecules that are packed together with no room to move, solids have a definite shape and volume. Liquids have molecules with some room to move, taking the shape of it's container while the volume remains the same. Gaseous matter have molecules so far apart that it has no set shape or volume but a gas can be compressed.
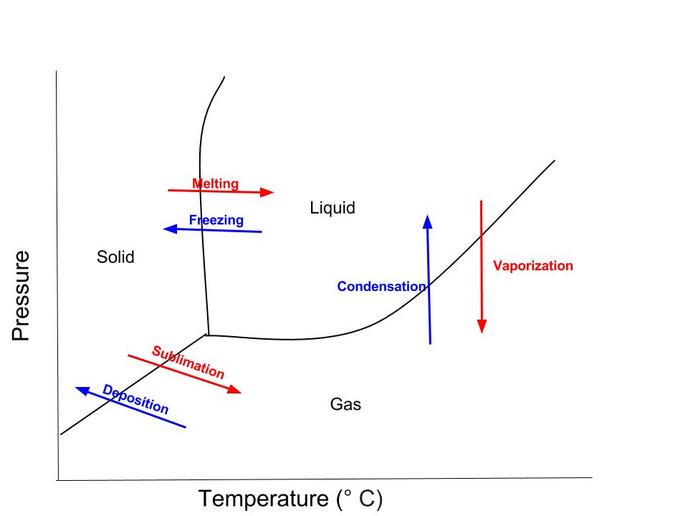
2. Draw a phase change diagram, and label all components.
3. What are the names and types of phases changes between the various phases?
There is freezing, melting, condensation, vaporization, sublimation and deposition. Liquids can freeze to become a solid or evaporate to become a gas. Solids melt to become a liquid, and sublimation occurs when it becomes a gas. Gases condense to become a liquid and deposition happens when gases become solid.
4. Explain how snow forms by deposition.
When the atmospheric temperature is at or below 0 degrees celsius and there is some moisture in the air. The snow will reach the ground if the ground is below freezing. When water vapor, or the gaseous state of water, from the cloud is cold (the atmospheric temperature) it turns into solid ice crystals.
5. Describe how liquid carbon dioxide can be achieved.
You can get liquid carbon dioxide if you increase the pressure of solid carbon dioxide. You increase the pressure to 5.1 atmospheres and keep the temperature below -20.5 degrees celsius. You put the carbon dioxide into a high pressure sealed container. The sublimation process increases the pressure forcing the carbon dioxide into a liquid form.
6. Describe what temperature and pressure combinations typically contribute to a solid, liquid, or gas being formed.
As the temperature rises, the faster the particles move. If it's a liquid, once it reaches high temperatures it becomes a gas. With low temperatures, it slows the particles down making a solid. Pressure only slightly reduces the space between closely packed molecules. Liquid often can't be compressed with pressure but the more pressure the more solid it becomes. High pressure and low temperatures often result in a solid, high temperatures and low pressure makes gas and midway between it becomes liquid.
7. Describe how to separate a mixture of two liquids.
There are two different types of liquids, immiscible and miscible. There are different separation techniques for these liquids and their different properties. Immiscible liquids (like oil and water) are easy to separate. You let the two liquids separate into two layers and let one liquid out. Miscible liquids however, dissolve into each other. I found a lot of different ways but fractional distillation is often used. Fraction distillation is possible with liquids of different boiling points. By boiling the mixture at different points it makes the different liquids evaporate. Or a centrifuge is used as well. It spins mixtures very quickly forcing the liquids with different densities to separate.
Solid matter has molecules that are packed together with no room to move, solids have a definite shape and volume. Liquids have molecules with some room to move, taking the shape of it's container while the volume remains the same. Gaseous matter have molecules so far apart that it has no set shape or volume but a gas can be compressed.
2. Draw a phase change diagram, and label all components.
3. What are the names and types of phases changes between the various phases?
There is freezing, melting, condensation, vaporization, sublimation and deposition. Liquids can freeze to become a solid or evaporate to become a gas. Solids melt to become a liquid, and sublimation occurs when it becomes a gas. Gases condense to become a liquid and deposition happens when gases become solid.
4. Explain how snow forms by deposition.
When the atmospheric temperature is at or below 0 degrees celsius and there is some moisture in the air. The snow will reach the ground if the ground is below freezing. When water vapor, or the gaseous state of water, from the cloud is cold (the atmospheric temperature) it turns into solid ice crystals.
5. Describe how liquid carbon dioxide can be achieved.
You can get liquid carbon dioxide if you increase the pressure of solid carbon dioxide. You increase the pressure to 5.1 atmospheres and keep the temperature below -20.5 degrees celsius. You put the carbon dioxide into a high pressure sealed container. The sublimation process increases the pressure forcing the carbon dioxide into a liquid form.
6. Describe what temperature and pressure combinations typically contribute to a solid, liquid, or gas being formed.
As the temperature rises, the faster the particles move. If it's a liquid, once it reaches high temperatures it becomes a gas. With low temperatures, it slows the particles down making a solid. Pressure only slightly reduces the space between closely packed molecules. Liquid often can't be compressed with pressure but the more pressure the more solid it becomes. High pressure and low temperatures often result in a solid, high temperatures and low pressure makes gas and midway between it becomes liquid.
7. Describe how to separate a mixture of two liquids.
There are two different types of liquids, immiscible and miscible. There are different separation techniques for these liquids and their different properties. Immiscible liquids (like oil and water) are easy to separate. You let the two liquids separate into two layers and let one liquid out. Miscible liquids however, dissolve into each other. I found a lot of different ways but fractional distillation is often used. Fraction distillation is possible with liquids of different boiling points. By boiling the mixture at different points it makes the different liquids evaporate. Or a centrifuge is used as well. It spins mixtures very quickly forcing the liquids with different densities to separate.
 RSS Feed
RSS Feed
